Istat test cartridges12/23/2023  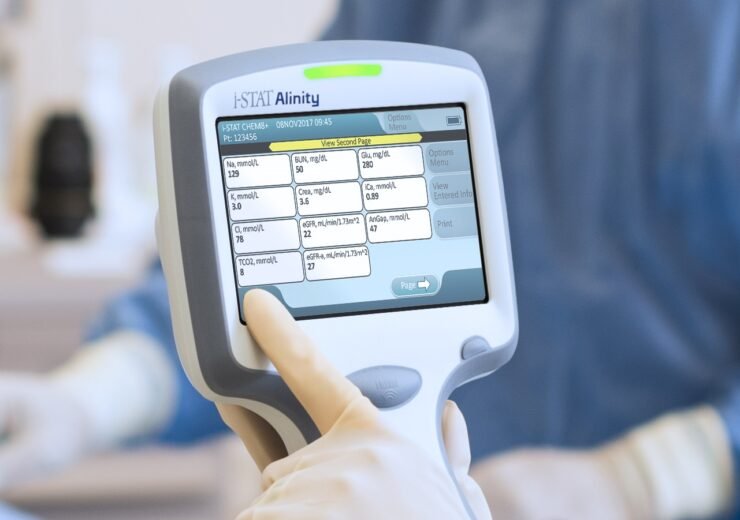
For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics. Check with your local representative for availability in specific markets. 
Not all products are available in all regions. GDPR Statement | Declaration for California Compliance Law. Any person depicted in such photographs is a model. Photos displayed are for illustrative purposes only. Two specialist commentators stated that the CHEM8+ and CG4+ cartridges do not include some of the additional tests that are used to help decide if a patient can be sent home safely, such as ddimer, troponin and white cell count, although cardiac troponin I is available on another iSTAT cartridge. Your use of this website and the information contained herein is subject to our Website Terms and Conditions and Privacy Policy. The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. This website is governed by applicable U.S. No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the company. Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. Both studies were supported by the manufacturer.©2023 Abbott. In the context of the overall service redesign, the contribution of the point‑of‑care testing to the reduced turnaround time cannot be evaluated separately. The i-STAT System delivers lab-quality results for a wide range of critical blood gas, electrolyte, chemistry,coagulation and cardiac marker tests in just. Both found a statistically significant reduction in the median time for patients to be declared ready to leave when the i‑STAT CHEM8+ and CG4+ cartridges were used. In the only UK‑based reports, 2 before‑and‑after studies assessed the impact of a redesigned emergency department service, including the addition of point‑of‑care testing using the i‑STAT, on the amount of time patients spent in the department. No statistically significant difference was found in time to intravenous antibiotics between the 2 groups. There was a statistically significant reduction in time to intravenous fluid administration in the i‑STAT group compared with the laboratory group. 
Eighty patients in the i‑STAT CG4+ group were compared with 80 patients in the laboratory group. One before‑and‑after study in a US emergency department compared bedside lactate testing using i‑STAT CG4+ with laboratory testing in patients with suspected sepsis. A prospective cohort study in 446 intubated intensive care adult patients found that partial pressure of oxygen and carbon dioxide as measured by the CG4+ cartridge could be considered acceptable surrogates to laboratory measurements. Test turnaround time was 2 minutes using the i‑STAT CG4+. Testing can be performed in 4 simple steps at the patient’s side with only 2 to 3 drops of whole blood. Each test cartridge has a unique combination of biosensors to suit a wide range of clinical needs. A before‑and‑after study found that post‑operative mortality rates in young children and babies changed from 6.2% before i‑STAT CG4+ lactate testing was introduced to 2.4% after. Single-use i-STAT test cartridges offer a broad menu of tests on a single, portable platform. Two studies evaluated the i‑STAT CG4+ cartridge in non‑emergency department settings. Of the 90 samples tested, the i‑STAT lactate values of 85 samples (94%) fell within the same risk category as the Vitros value. The Vitros system was used as a reference standard. User did not respond to mandatory prompt before handheld time out. One prospective cohort study in a US medicine and pathology laboratory assessed the correlation between 5 different systems used for lactate analysis, including the i‑STAT CG4+ cartridge. Analysis showed that the lactate measurement using the i‑STAT was accurate for clinical decision‑making compared with the laboratory test. Five used the CG4+ cartridge, 1 used the CHEM8+ cartridge and the other used both CG4+ and CHEM8+ cartridges.Ī prospective cohort study in 1 US emergency department assessed the diagnostic test accuracy of lactate in serum measured using the CG4+ cartridge for predicting mortality in 669 patients with suspected sepsis. Seven studies using i‑STAT with findings relevant to the emergency department setting are included in this briefing. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
